TABLE OF CONTENTS
What Are the FDA Supplement Facts Label Guidelines?
What Must a Supplement Facts Label Include?
- Serving Size and Servings Per Container
- Nutrient Declaration
- Amounts Per Serving
- The % Daily Value
- Other Dietary Ingredients
- Other Ingredients (Inactive)
- The Name and Place of Business of the Manufacturer
- Net Quantity of Contents
- Accuracy of Declared Amounts
FDA Supplement Facts Label Format Requirements
The Panel Must Be Enclosed in a Box
The Title "Supplement Facts" Must Stand Out
When You Have Multiple Serving Options
Products Sold From Bulk Containers
Supplement Facts Label vs. Nutrition Facts Label: Key Differences
What Ingredients Can Be Listed
Who Is Exempt From Supplement Facts Label Requirements?
FAQs About Supplement Facts Label Requirements
- What must a supplement facts label include by law?
- What are the FDA requirements for supplement facts labels?
- What is the difference between a nutrition facts label and a supplement facts label?
- What font size and format does the FDA require on supplement facts labels?
- Are all dietary supplements required to have a supplement facts label?
If you've ever launched a dietary supplement or thought about getting into the supplement business, you already know there's a lot more to it than creating a great product. Before that product ever reaches a customer's hands, the label has to be right, and by "right," we mean compliant with strict FDA guidelines.
There are specific rules around what information must be included in the supplement facts label. There are also rules about how it should be formatted, which font size to use, and where everything needs to appear on the packaging.
In this article, we'll walk you through everything you need to know about supplement facts label requirements, straight from the FDA guidelines, so you can stay compliant, build consumer trust, and avoid costly mistakes.
What Are the FDA Supplement Facts Label Guidelines?
The FDA supplement facts label guidelines are rooted in the Dietary Supplement Health and Education Act (DSHEA) of 1994, which gave the FDA authority to regulate dietary supplements as a distinct category from conventional food and drugs.
Since then, the FDA has issued detailed regulations (primarily under 21 CFR Part 101) that spell out exactly what a supplement label must look like and what it must say.
The main idea behind the supplement facts label FDA regulations, is that consumers have a right to know what they're taking into their body. The label is the primary way that information gets communicated, which is why the FDA takes compliance so seriously.
It's important to understand that, unlike pharmaceutical drugs, dietary supplements don't require FDA approval before they go to market. However, that doesn't mean a brand can wake up and release any supplement into the market. Supplement facts label FDA guidelines are still legally binding, and manufacturers are fully responsible for ensuring their labels meet every requirement before a product is sold.
So what exactly do these guidelines cover?
The supplement facts label guidelines cover the specific information that must appear on the label, the order in which it's presented, the formatting and font size, and even the exact wording required in certain sections.
We'll explain all of that in the sections below.
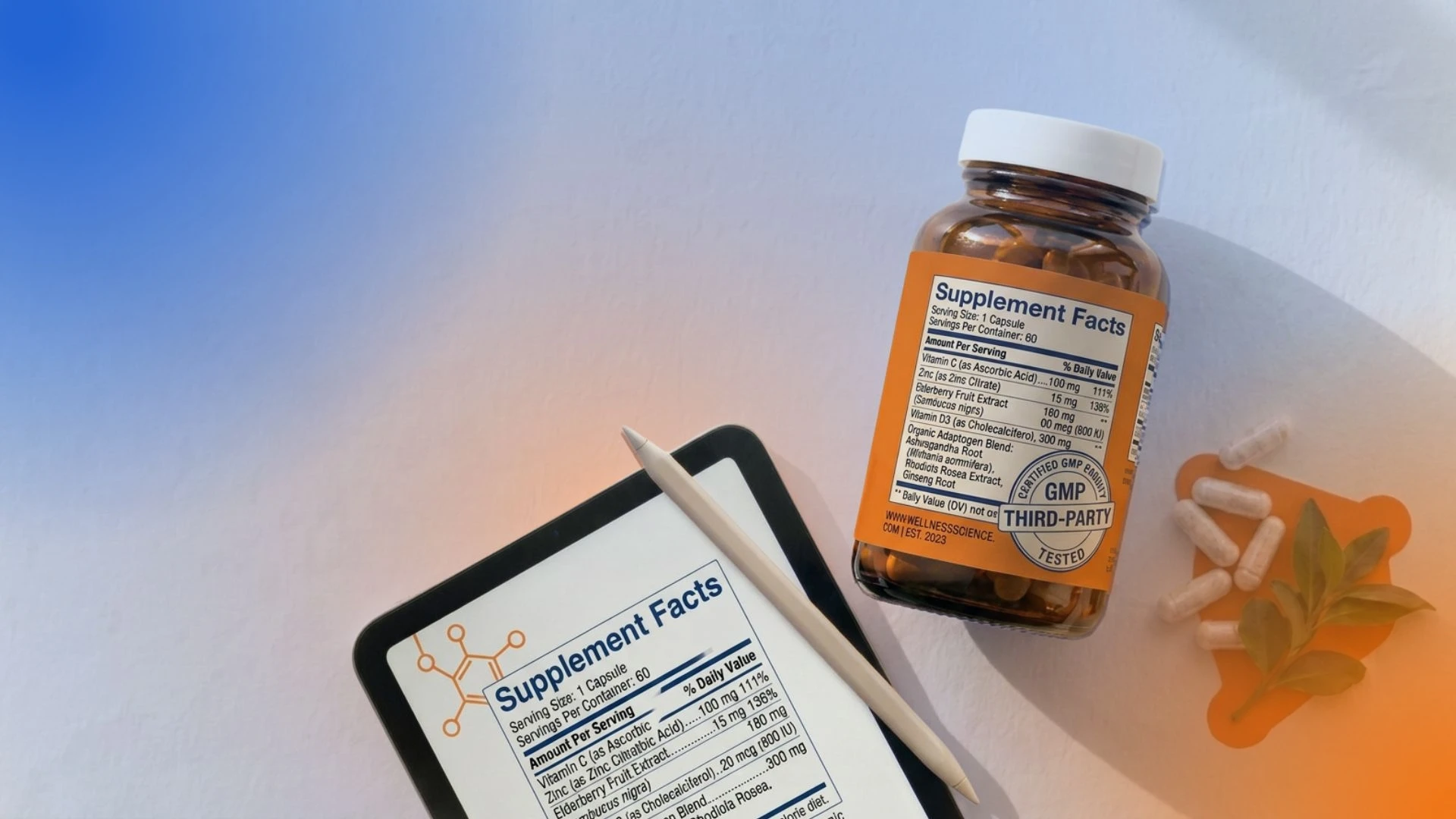
What Must a Supplement Facts Label Include?

According to FDA supplement facts label guidelines, the supplement facts label must include the following information:
1. Serving Size and Servings Per Container
The serving size is the first thing listed on the panel, and the FDA is strict about how it's written. One serving of a dietary supplement equals the maximum amount recommended on the label for consumption.
So if your label directions say to take one to three tablets with breakfast, the serving size would be three tablets. And unlike food labels, there is no flexibility here; the FDA requires you to use the exact term "Serving Size." No alternatives are permitted.
Servings per container must also be listed, unless the information is already captured in the net quantity of contents statement. For example, if your label says the bottle contains 100 tablets and the serving size is one tablet, listing "Servings Per Container: 100" would be redundant and isn't required.
2. Nutrient Declaration
The supplement facts label must declare certain nutrients whenever they are present in measurable amounts. These include total calories, calories from fat, total fat, saturated fat, cholesterol, sodium, total carbohydrate, dietary fiber, sugars, protein, vitamin A, vitamin C, calcium, and iron. Trans fat must also be listed on a separate line beneath saturated fat when present in measurable amounts.
Aside from these mandatory nutrients, other nutrients such as polyunsaturated fat, monounsaturated fat, soluble fiber, and sugar alcohols may be voluntarily declared. However, you must declare them if you make any claims about them on the label.
Vitamins and minerals, excluding the four major ones (vitamin A, vitamin C, calcium, and iron), only need to be listed if they have been added for the purpose of supplementation, or if you make a claim about them. So if vitamin E occurs naturally in your product and you make no claim for it, you are not required to list it.
Dietary ingredients must also be listed in a specific order. For vitamins and minerals with established daily values, the FDA requires them to appear in the same order as conventional food labels, with vitamins and minerals grouped together, starting with Vitamin A and ending with potassium.
3. Amounts Per Serving
Every dietary ingredient listed on the supplements facts label must include the quantitative amount per serving.
For nutrients gotten from a compound like calcium from calcium carbonate, you must list the weight of the actual nutrient (calcium), not the source ingredient (calcium carbonate).
Amounts can be placed in a separate column or immediately after the ingredient name, and the heading "Amount Per Serving" can be replaced with equivalent language such as "Each Tablet Contains" or "Amount Per 2 Tablets."
4. The % Daily Value
For every dietary ingredient that has an established daily value, the label must display the percent daily value (%DV).
You calculate this by dividing the nutrient amount per serving by the established daily value, then multiplying by 100. Percentages must be rounded to the nearest whole number; if the amount is present but rounds to 0%, the label must state "Less than 1%" rather than showing 0%.
For ingredients with no established daily value (which is common with herbal extracts and many other dietary ingredients), you must place a symbol in the %DV column that points to the footnote "Daily Value Not Established."
5. Other Dietary Ingredients
Dietary ingredients that don't have daily values, like herbs, botanicals, amino acids, and certain enzymes, must be listed separately in the panel, following the nutrients that do have daily values.
These must be listed by their common or usual name, along with the quantitative amount per serving. The FDA does not specify a required order for these ingredients.
If your product contains a proprietary blend (a combination of ingredients in supplements, foods, or cosmetics where the total weight of the blend is listed, but the specific amounts of individual ingredients are hidden), the label must identify it as a "Proprietary Blend" or use a descriptive name, list the total weight of all ingredients in the blend on the same line, and then list each ingredient within the blend in descending order of predominance by weight, indented beneath the blend name.
6. Other Ingredients (Inactive)
Ingredients that are not dietary ingredients, like binders, fillers, excipients, coatings, and flavoring agents, must be listed in a separate "Other Ingredients" statement below the supplement facts panel, in descending order of predominance by weight.
These are not listed inside the panel itself. It's worth noting that if you use a magnesium salt as a binder, for example, it belongs in this ingredient statement, not in the supplement facts panel.
7. The Name and Place of Business of the Manufacturer
The label must include the name and address of the manufacturer, packer, or distributor.
This gives consumers and regulators a point of contact if there are any questions or concerns about the product.
8. Net Quantity of Contents
The label must state the total amount of product in the container, for example, "60 capsules" or "300g."
This helps consumers understand what they're getting and compare products accurately.
9. Accuracy of Declared Amounts
For dietary ingredients that have been specifically added to a product, the supplement must contain 100% of the declared amount.
For naturally occurring dietary ingredients, the product must contain at least 80% of the declared value. If you fall short of these thresholds, your product is considered misbranded and in violation of federal law.
FDA Supplement Facts Label Format Requirements
The supplement facts label FDA guidelines, also have very specific rules about how the panel must look and be formatted.
If you get the information right, but present it incorrectly, you would still have a non-compliant label.
Here's what the supplement facts label FDA format rules require:
The Panel Must Be Enclosed in a Box
The entire supplement facts panel must be enclosed in a box using hairlines (thin ruled lines that separate the panel from the rest of the label).
Inside the panel, a hairline rule must be centered between the lines of text to separate each dietary ingredient from the one above and below it.
There are exceptions for small and intermediate-sized packages, which we'll explain below.
The Title "Supplement Facts" Must Stand Out
The title "Supplement Facts" must be larger than all other print in the panel and must be set to the full width of the panel.
The title and all headings must be bolded to distinguish them from the rest of the information. This is a non-negotiable part of the supplement facts label guidelines.
No other text in the panel should be as prominent as the title itself.
Type Size Requirements
According to FDA supplement facts label guidelines, all information in the panel, other than the title, headings, and footnotes, must be set in a uniform type size of no smaller than 8 point.
Column headings and footnotes may be set in no smaller than 6 point. The supplement label font size requirements are slightly different for smaller packaging, which the FDA categorizes as follows:
- Small packages (less than 12 square inches of total label surface area) may use type as small as 4.5 point
- Intermediate-sized packages (12 to 40 square inches) must use type no smaller than 6 point, though 4.5 point is permitted under certain conditions. For example, when a package between 20 and 40 square inches lists more than 16 dietary ingredients
Type Style and Presentation
The supplement facts label FDA guidelines require that all information be presented in a single, easy-to-read type style.
The text must be printed in all black or a single color on a white or neutral contrasting background.
Upper and lowercase letters must be used, though all uppercase is permitted on small packages. Letters must not touch each other, and at least one point of leading (space between lines of text) is required.
Tabular and Linear Formats
The standard format for a supplement facts panel is a vertical column layout. However, for small and intermediate-sized packages with limited space, the FDA permits a tabular format.
If even that won't fit, a linear (string) format may be used, where all information is presented on a single continuous line.
These alternative formats exist to ensure that supplement facts label FDA compliance is achieved regardless of packaging size.
When You Have Multiple Serving Options
If your product contains two or more packets with different serving sizes, for example, a morning capsule and an evening capsule, you may present the information for each packet in individual panels or on an aggregate label.
An aggregate label for two packets would use five columns: one for ingredient names, two for the morning packet (amounts and %Daily Value), and two for the evening packet.
Where the Panel Can Be Placed
Under FDA supplement facts label guidelines, the panel is placed on the side of the packaging to the right of the display panel. But, if there isn't enough space there, it may be placed on any other panel that consumers can readily see.
For products sold in multi-unit retail packs, the supplement facts panel may be omitted from individual unit containers, as long as it appears fully on the outer package and each individual unit is labeled with the statement "This Unit Not Labeled For Retail Sale."
Products Sold From Bulk Containers
If your dietary supplements are sold from bulk containers rather than in individual packaging, the retailer is required to clearly display the supplement facts panel at the point of purchase (on a counter card, sign, tag, or similar display).
Alternatively, the information may be made available in a booklet or binder at the point of purchase.
NOTE: A label that includes all the required information but fails to meet the FDA's presentation standards is still considered non-compliant, and that's a risk no supplement brand should be willing to take.
Supplement Facts Label vs. Nutrition Facts Label: Key Differences
One of the most common points of confusion for new supplement brands is understanding how a dietary supplement facts label is different from the nutrition facts label found on conventional food products.
Here's a breakdown of the key differences:
The Title
The most obvious difference is the title itself. Food products carry a "Nutrition Facts" label.
Dietary supplements carry a "Supplement Facts" label.
What Ingredients Can Be Listed
On a supplement facts label, you are required to list dietary ingredients that don't have established Reference Daily Intakes (RDIs) or Daily Reference Values (DRVs). This includes things like herbs, botanicals, and certain amino acids.
You are not permitted to list these types of ingredients on a nutrition facts label for conventional food.
Source of Dietary Ingredients
On a supplement facts label, you may list the source of a dietary ingredient. For example, "Vitamin C (from rose hips)." This is not permitted on a nutrition facts label for food.
Also, for herbal ingredients, the supplement facts label must identify the part of the plant from which the ingredient is derived (e.g., root, leaf, stem). This level of detail is not required or permitted on food labels.
Zero Amounts
On a nutrition facts label, you are required to list zero amounts for mandatory nutrients, even if none of that nutrient is present.
On a dietary supplement facts label, you are not permitted to list zero amounts. If a nutrient is not present in a measurable amount, you put <1, or it simply doesn't appear on the supplements facts label at all.
The Inactive Ingredients List
On a supplement label, the inactive ingredients list like binders, fillers, coatings, and excipients, must appear in a separate "Other Ingredients" statement below the supplement facts panel, not inside it.
This is different from food labeling, where all ingredients appear together in a single ingredient list.
Font Size and Formatting
The supplement label font-size requirements include specific provisions for smaller supplement packaging that don't apply to standard food labels.
The FDA supplement facts label template is also visually different from the nutrition facts layout, particularly in how "other dietary ingredients" without daily values are presented.
The % Daily Value Footnote
On conventional food labels, a footnote comparing a 2,000-calorie diet to a 2,500-calorie diet is required.
On a supplement facts label, this footnote is not required. However, if your supplement declares total fat, saturated fat, total carbohydrate, dietary fiber, or protein, you are still required to include the footnote: "Percent Daily Values are based on a 2,000 calorie diet."
Who Is Exempt From Supplement Facts Label Requirements?

Your product may be exempt from the dietary supplement facts label requirement if your business meets one of the following criteria:
- Small businesses: If your firm has no more than $50,000 in gross sales of food to consumers, or no more than $500,000 in total annual sales, you may qualify for an exemption
- Low-volume products: If your firm sells fewer than 100,000 units of the product per year and has fewer than 100 full-time equivalent employees, you may be eligible, provided you file an annual notification with the FDA
- Bulk shipments: If your product is shipped in bulk form and is not distributed directly to consumers in that form, and is instead supplied for use in the manufacture of other dietary supplements, it may be exempt
It's important to note that both the small business and low-volume exemptions are only available if your product label carries no claims or other nutrition information.
The moment you make any kind of nutrient content claim or health-related claim on the label, the exemption no longer applies.
Wrapping Up
The FDA supplement facts label guidelines exist to ensure that your consumers know exactly what they're buying, and if you take those guidelines seriously, you’ll build lasting credibility in the market.
When you’re unsure of the rules, refer directly to the supplement facts label FDA regulations under 21 CFR Part 101, or work with a compliance specialist who knows the rules.
Your label is the first thing a customer sees. So make sure it says the right things in the right way.
FAQs About Supplement Facts Label Requirements
1. What must a supplement facts label include by law?
According to FDA supplement facts label guidelines, the supplement facts label must include the serving size, servings per container, the name and quantity of every dietary ingredient, the percent daily value for nutrients with established daily values, an "Other Ingredients" statement for inactive ingredients, and the name and address of the manufacturer or distributor.
The label must also state the net quantity of contents and comply with all formatting requirements set out under 21 CFR Part 101.
2. What are the FDA requirements for supplement facts labels?
The FDA supplement facts label guidelines cover both content and presentation. On the content side, manufacturers must list all dietary ingredients, their amounts per serving, and the %DV where applicable. On the formatting side, the panel must be enclosed in a box using hairlines, the title "Supplement Facts" must be the largest text in the panel, and all body text must be set in a uniform type size of no smaller than 8 point. The supplement facts label FDA regulations also specify how proprietary blends, herbal extracts, and inactive ingredients must be disclosed.
3. What is the difference between a nutrition facts label and a supplement facts label?
A dietary supplement facts label must include dietary ingredients without established daily values, such as herbs and botanicals, which are not permitted on a nutrition facts label. The supplement facts label also allows the source of a dietary ingredient to be listed, requires the part of the plant to be identified for herbal ingredients, and does not permit the listing of zero amounts. The inactive ingredients list on a supplement label also appears separately below the panel, rather than within it.
4. What font size and format does the FDA require on supplement facts labels?
For standard packaging, the supplement label font size requirements specify that all body text in the panel must be set in a uniform type size of no smaller than 8 point, while column headings and footnotes may be as small as 6 point. For small packages (those with less than 12 square inches of label surface area), a minimum of 4.5 point is permitted.
For intermediate-sized packages, the minimum is generally 6 point, with 4.5 point allowed under specific conditions. The FDA supplement facts label template also requires a single, easy-to-read type style, with text printed in black or a single color on a white or neutral background.
5. Are all dietary supplements required to have a supplement facts label?
Not all products are required to carry a supplement facts label. Small businesses with under $50,000 in consumer food sales, low-volume products sold by companies with fewer than 100 full-time employees and fewer than 100,000 units sold annually, and bulk shipments intended for use in manufacturing other supplements may qualify for an exemption. However, these exemptions are voided the moment any nutrition or health claim appears on the label.





.svg)

.avif)


.svg)




